by Bruce Wells | Mar 20, 2024 | Petroleum Products
Popular but dangerous mixture replaced by brighter, less volatile lamp fuel.
In the early 19th century, lamp designs burned many different fuels, including rapeseed oil, lard, and whale oil rendered from whale blubber (and the more expensive spermaceti from the heads of sperm whales), but most Americans could only afford light emitted by animal-fat, tallow candles.
By 1850, the U.S. Patent Office recorded almost 250 different patents for all manner of lamps, wicks, burners, and fuels to meet growing consumer demand for illumination. At the time, most Americans still lived in almost complete darkness when the sun went down. (more…)
by Bruce Wells | Feb 21, 2024 | Petroleum Products
Revolutionary DuPont lab product first used commercially in 1938 for toothbrush bristles.
The world’s first synthetic fiber was the petroleum product “Nylon 6,” discovered in 1935 by a DuPont chemist who produced the polymer from chemicals found in oil.
DuPont Corporation foresaw the future of “strong as steel” artificial fibers. The chemical conglomerate had been founded in 1802 as a Wilmington, Delaware, manufacturer of gunpowder. The company would become a global giant after its scientists created durable and versatile products like nylon, rayon and lucite.

“Women show off their nylon pantyhose to a newspaper photographer, circa 1942,” noted historian Jennifer S. Li in “The Story of Nylon – From a Depressed Scientist to Essential Swimwear.” Photo by R. Dale Rooks (1917-1954).
The world’s first synthetic fiber — nylon — was discovered on February 28, 1935, by a former Harvard professor working at a DuPont research laboratory. Called Nylon 6 by scientists, the revolutionary carbon-based product came from chemicals found in petroleum.
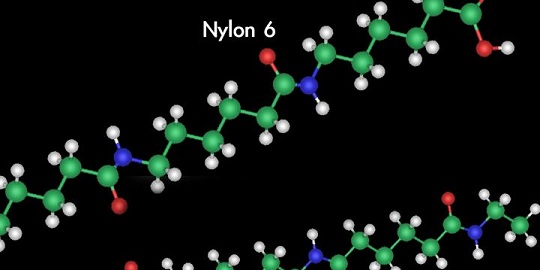
Chemists called the man-made fiber Nylon 6 because chains of adipic acid and hexamethylene diamine each contained six carbon atoms per molecule.
Professor Wallace Carothers had experimented with artificial materials for more than six years. He previously discovered neoprene rubber (commonly used in wet suits) and made major contributions to understanding polymers — large molecules composed in long chains of repeating chemical structures.
Polymer Chains
Carothers, 32, created fibers when he combined the chemicals amine, hexamethylene diamine, and adipic acid. His experiments formed polymer chains using a process in which individual molecules joined together with water as a byproduct. But the fibers were weak.
A PBS series, A Science Odyssey: People and Discoveries, in 1998 noted Carothers’ breakthrough came when he realized, “the water produced by the reaction was dropping back into the mixture and getting in the way of more polymers forming. He adjusted his equipment so that the water was distilled and removed from the system. It worked!”

DuPont named the petroleum product nylon — although chemists called it Nylon 6 because the adipic acid and hexamethylene diamine each contain six carbon atoms per molecule.

“Until now, all good toothbrushes were made with animal bristles,” noted a 1938 ad.
Each man-made molecule consists of 100 or more repeating units of carbon, hydrogen, and oxygen atoms, strung in a chain. A single filament of nylon may have a million or more molecules, each taking some of the strain when the filament is stretched.
There’s disagreement about how the product name originated at DuPont.
“As to the word nylon, it’s actually quite arbitrary. DuPont itself has stated that originally the name was intended to be No-Run (that’s run as in the sense of the compound chain of the substance unravelling), but at the time there was no real justification for the claim, so it needed to be changed,” noted Chris Nickson in a 2017 website post, Where Does the Name Nylon Originate?
Toothbrush Bristles
The first commercial use of this revolutionary petroleum product was for toothbrushes.
On February 24, 1938, the Weco Products Company of Chicago, Illinois, began selling its new “Dr. West’s Miracle-Tuft” — the earliest toothbrush to use synthetic DuPont nylon bristles.

First used for toothbrush bristles, nylon women’s stockings were promoted in a DuPont 1948 ad.
Americans will soon brush their teeth with nylon — instead of hog bristles, declared an article in the New York Times. “Until now, all good toothbrushes were made with animal bristles,” explained a 1938 Weco Products advertisement in Life magazine.
“Today, Dr. West’s new Miracle-Tuft is a single exception,” the ad proclaimed. “It is made with EXTON, a unique bristle-like filament developed by the great DuPont laboratories, and produced exclusively for Dr. West’s.”

Pricing its toothbrush at 50 cents, the Weco Products Company guaranteed, “no bristle shedding.” Johnson & Johnson of New Brunswick, New Jersey, will introduce a competing nylon-bristle toothbrush in 1939.
Nylon Stockings
Although DuPont patented nylon in 1935, it was not officially announced to the public until October 27, 1938, in New York City.
A DuPont vice president unveiled the synthetic fiber — not to a scientific society or industry association — but to 3,000 Women’s Club members gathered at the site of the upcoming 1939 New York World’s Fair.

During WWII, nylon was used as a substitute for silk in parachutes.
“He spoke in a session entitled ‘We Enter the World of Tomorrow,’ which was keyed to the theme of the forthcoming fair, the World of Tomorrow,” explained DuPont historian David A. Hounshell in a 1988 book.
The petroleum product was an instant hit, especially as a replacement for silk in hosiery. DuPont built a full-scale nylon plant in Seaford, Delaware, and began commercial production in late 1939.
The company purposefully did not register “nylon” as a trademark – choosing to allow the word to enter the American vocabulary as a synonym for “stockings.”
Women’s nylon stockings appeared for the first time at Gimbels Department Store on May 15, 1940. World War II would remove the polymer hosiery to make nylon parachutes and other vital supplies.

Nylon would become far and away the biggest money-maker in the history of DuPont. The powerful material from lab research led company executives to derive formulas for growth, according to Hounshell in The Nylon Drama.
“By putting more money into fundamental research, Du Pont would discover and develop ‘new nylons,’ that is, new proprietary products sold to industrial customers and having the growth potential of nylon,” Hounshell explained in his 1988 book.
Carothers did not live to see the widespread application of his work — in consumer goods such as toothbrushes, fishing lines, luggage and lingerie, or in special uses such as surgical thread, parachutes, or pipes — nor the powerful effect it had in launching a whole era of synthetics.
Devastated by the sudden death of his favorite sister in early 1937, Carothers committed suicide in April of that year. The DuPont Company would name its research facility after him.
As the DuPont website notes, the invention of nylon radically changed the way people dressed worldwide — and rendered the term ‘silk stocking’ obsolete (and once an epithet directed at the wealthy elite).
Nylon’s success encouraged DuPont to adopt long-term strategies for products developed from basic research.
_______________________
Recommended Reading: The Golden Thread: How Fabric Changed History (2019); Enough for One Lifetime: Wallace Carothers, Inventor of Nylon (2005); The Nylon Drama (1988). Your Amazon purchases benefit the American Oil & Gas Historical Society; as an Amazon Associate, AOGHS earns a commission from qualifying purchases.
_______________________
The American Oil & Gas Historical Society (AOGHS) preserves U.S. petroleum history. Become an AOGHS annual supporting member and help maintain this energy education website and expand historical research. For more information, contact bawells@aoghs.org. © 2023 Bruce A. Wells. All rights reserved.
Citation Information – Article Title: “Nylon, a Petroleum Polymer.” Authors: B.A. Wells and K.L. Wells. Website Name: American Oil & Gas Historical Society. URL: https://aoghs.org/products/petroleum-product-nylon-fiber. Last Updated: February 21, 2024. Original Published Date: February 23, 2014.
View Post
by Bruce Wells | Dec 15, 2023 | Petroleum Products
Petroleum product at bottom of refining process improved American mobility.
As the U.S. centennial neared, President Ulysses S. Grant directed that Pennsylvania Avenue be paved with Trinidad asphalt. By 1876, the president’s paving project covered about 54,000 square yards, according to the National Asphalt Pavement Association.

Pennsylvania Avenue was first paved in 1876-77 with bitumen imported from Trinidad. Thirty-one years later, a better asphalt derived from petroleum distillation was used to repave the famed pathway to the Capitol, above. Photo courtesy the Asphalt Institute.
“Brooms, lutes, squeegees and tampers were used in what was a highly labor intensive process. Only after the asphalt was dumped, spread, and smoothed by hand did the relatively sophisticated horse-drawn roller, and later the steam roller, move in to complete the job,” NAPA noted in its 1992 publication, A Century of Progress: The History of Hot Mix Asphalt. (more…)
by Bruce Wells | Dec 14, 2023 | Petroleum Products
Petroleum paraffin soon found its way into candles, crayons, chewing gum…and a peculiar wax candy.
When Ralphie Parker and his 4th-grade classmates dejectedly handed over their Wax Fangs to Mrs. Shields in “A Christmas Story,” a generation might be reminded of what a penny used to buy at the local Woolworth’s store. But there is far more to these paraffin playthings than a penny’s worth of fun.
It’s hard to recall a time when there were no Wax Lips, Wax Moustaches, or Wax Fangs for kids to smuggle into classrooms. Many grownups may remember the peculiar disintegrating flavor of Wax Lips from bygone Halloweens and birthday parties, but few know where these enduring icons of American culture actually started.
The answer can be found by way of the oil patch.
Beginning with the August 1859 first commercial U.S. oil well, Pennsylvania oilfields quickly brought an important new source for refining kerosene. “This flood of American petroleum poured in upon us by millions of gallons, and giving light at a fifth of the cost of the cheapest candle,” wrote British chandler James Wilson in 1879.

Released on November 18, 1983, “A Christmas Story” featured Ralphie, his 4th-grade classmates – and a popular petroleum product. Photos courtesy MGM Home Entertainment.
As kerosene lamps replaced candles for illumination, the much-reduced candle business turned from tallow to versatile paraffin.
A byproduct of kerosene distillation, paraffin found its way from refinery to marketplace in candles, sealing waxes and chewing gums. Ninety percent of all candles by 1900 used paraffin as the new century brought a host of novel uses. Thomas Edison’s popular new phonographs also needed paraffin for their wax cylinders.

Crayons were introduced by the Binney & Smith Company in 1903 and were instantly successful. Alice Binney came up with the name by combining the French word for chalk, craie, with an English adjective meaning oily, oleaginous: Crayola (see Carbon Black and Oilfield Crayons).

Concord Confections, part of Tootsie-Roll Industries, continues to produce Wax Lips and other paraffin candies for new generations of schoolchildren.
In New York City, after collecting unrefined waxy samples from Pennsylvania oil wells, Robert Chesebrough invented a method for turning paraffin into a balm he called “petroleum jelly,” later “Vaseline.” His product also led to a modern cosmetic giant (learn more in The Crude History of Mabel’s Eyelashes).
Paraffin Lips, Fangs, and Horses Teeth
An inspired Buffalo, New York, confectioner soon used fully refined, food-grade paraffin and a sense of humor to find a niche in America’s imagination.
When John W. Glenn introduced children to paraffin “penny chewing gum novelties,” his business boomed. By 1923, his J.W. Glenn Company employed 100 people, including 18 traveling sales representatives.
Glenn Confections became the wax candy division of Franklin Gurley’s nearby W.&F. Manufacturing Company. There, the ancestors of Wax Lips chattered profitably down the production line. Among the most popular of these novelties at the time were Wax Horse Teeth (said to taste like wintergreen).
By 1939, Gurley was producing a popular series of holiday candles for the Socony-Vacuum Oil Company using paraffin from a nearby refinery at Olean, New York — once home to the world’s largest crude oil storage site. A field of metal tanks, some holding 20,000 gallons of paraffin, stood next to Gurley’s W.&F. Manufacturing Company in Buffalo.

Glenn Confections, the candy division of W. & F. Manufacturing Company, produced Fun Gum Sugar Lips, Wax Fangs, and Nik-L-Nips.
Decorative and scented paraffin candles soon became the company’s principal products, accounting for 98 percent of W.&F. Manufacturing sales. Gurley’s “Tavern Candle” Santas, reindeer, elves and other colorful Christmas favorites today are prized by collectors on eBay, as are his elaborately molded Halloween candles.
As W.&F.’s wax candy division, Glenn Confections, has continued to manufacture Fun Gum Sugar Lips, Wax Fangs, and the small, wax bottles — Nik-L-Nips available from the Old Time Candy Company.

In Emlenton, Pennsylvania, a few miles south of Oil City, the Emlenton Refining Company (and later the Quaker State Oil Refining Company) provided the fully refined, food-grade paraffin for these bizarre but beloved treats.
Retired Quaker State employee Barney Lewis remembers selling Emlenton paraffin to W.&F. Manufacturing.
During a 2005 interview, Lewis noted, “It was always fun going to the plant…they were very secret about how they did stuff, but you always got a sample to bring home,” adding, “Wax Lips, Nik-L-Nips…the little Coke bottle-shaped wax, filled with colored syrup.”
Concord Confections, a small part of Tootsie-Roll Industries, continues to produce Wax Lips and other paraffin candies for new generations of schoolchildren. The modern petroleum industry produces an astonishing range of products for consumers. But among the many products that find their history in the oilfield, few are as unique and peculiar as Wax Lips.
In December 2007, “A Christmas Story” was ranked the number one Christmas film of all time by AOL. Set in 1940, the movie has been shown in an annual marathon since 1997.
Among the waxy petroleum products featured is a polymer “major award” — the plastic leg-lamp with the black nylon stocking.
_______________________
Recommended Reading: Sweet!: The Delicious Story of Candy (2009); How Sweet It Is (and Was): The History of Candy (2003). Your Amazon purchase benefits the American Oil & Gas Historical Society. As an Amazon Associate, AOGHS earns a commission from qualifying purchases.
_______________________
The American Oil & Gas Historical Society preserves U.S. petroleum history. Become an AOGHS annual supporting member and help maintain this energy education website and expand historical research. For more information, contact bawells@aoghs.org. Copyright © 2023 Bruce A. Wells. All rights reserved.
Citation Information – Article Title: “Oleaginous History of Wax Lips.” Authors: B.A. Wells and K.L. Wells. Website Name: American Oil & Gas Historical Society. URL: https://aoghs.org/products/an-oleaginous-history-of-wax-lips. Last Updated: December 15, 2022. Original Published Date: December 1, 2006.












